 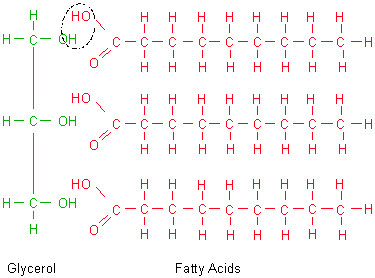
If you were to drink a solution of glycerol it will actually be sweet. We see why it's an alcohol, it's got its hydroxyl groups. And this is considered to be a sugar alcohol. It has three hydroxyl groups right over here. If you just get attached to one of them, this makes you an alcohol so this definitely makes it an alcohol. Each of those carbons is attached to a hydroxyl group, an O-H group. We can see we have a three carbon chain right over here. Let me do it in that same blue that I just wrote glycerol in. This molecule on the left, this is glycerol. And so here I have the constituent molecules of a triglyceride. But let's think about what a triglyceride is. If you were to get your cholesterol checked or if your parents get their cholesterol checked, they'll probably get a report on triglycerides which is really a measure of the fat concentration in their blood. Or where what most people typically refer to as fat, they're talking about triglycerides. But triglycerides, this is literally fat. I actually like this second name more because it makes sense when you break down the molecule, triacylglycerol. But it can also be referred to as tri- And I'm going to do the different parts in different colors. And a more technical term for triglycerides- Triglyceride is the word that you might hear when you go to the doctor's office. Fats in a liquid form is sometimes referred to as oils. Which is often kind of a feared word but as we'll see it's essential for life. And in everyday language we often call these fats. Let's study the molecular structure of triglycerides. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
